Volunteer for a Clinical Research Study in California and Save Lives!
What are Clinical Trials?
Clinical trials are research studies performed on humans to help evaluate a new medical treatment, drug or device. The main purpose of a clinical trial is to discover new and improved methods for treating and preventing different diseases, illnesses, and more.
Featured Clinical Research Study
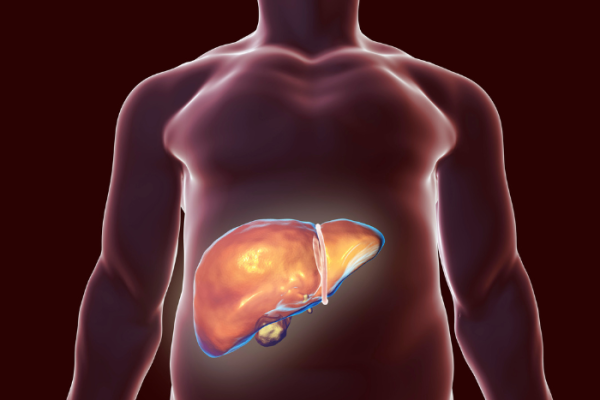
NASH Study
FOMAT is enrolling participants for a NASH Clinical Trial.
Non-alcoholic steatohepatitis (NASH) is a silent disease that often stays asymptomatic until advanced stages. It involves excess liver fat, leading to inflammation and damage over time. In early phases, it goes unnoticed, but as NASH advances, it can become cirrhosis, severely impairing liver function and risking complications. Early detection and action are vital to prevent NASH progression and protect liver health.
Active Studies
Upcoming Studies
Study Types Availables
Make an Impact
Join One of Our Clinical Research Studies Today
Frequently Asked Questions
How much time and effort does it take?
The length of a clinical trial varies depending on several factors, such as the type of illness and the type of medicine being researched. For example, if the study is researching a treatment for a chronic condition, participants’ progress on the trial may be tracked over a relatively long period of time. Studies of antibiotics on the other-hand conclude quickly as patients are typically treated on study for a few days only.
Source: Novartis
Do I have to track my progress?
Depending on the study and the study process, in some cases, you will have to keep track of your process, in other cases you will not, but you will have the support and guidance of the research team.
Can I drop out of the study at any time?
As a research participant, you have the right to drop out of a study at anytime, the right to be given new information about the study, and the right to ask questions at anytime and have them answered promptly.
Do I have to provide my medical history?
Yes. Although the purpose of the study is not to treat an individual for any health condition, a study may involve clinical procedures such as an MRI, blood tests, hearing tests, etc. In order to complete these and other tests, the researcher must know the volunteer's general state of health to ensure his or her safety during the clinical study process.
Do I have to be a US citizen?
No, you do not have to be a US citizen to participate in a clinical trial. We also do not ask for your social security number. All we ask is that you have a physical address and some form of ID that we can use during the enrollment process.
Can I invite others to join the study with me?
Sure, you can refer any friend or family member who is willing to participate in a study, for more information click here!




